I have a collaboration with my Surrey colleague Joe Keddie to study these systems. I run the modelling side of the collaboration, he runs the experimental side.
Rubber gloves
Rubber gloves are made by dipping a hand-shaped mould covered with a salt into a liquid containing small particles. The salt dissolves in the liquid, causing the liquid near the surface of the mould to have a high salt concentration. This in turn causes the particles to stick together forming a gel layer in contact with the mould. The mould is then pulled out of the liquid, and the gel layer dries to form the glove.
We have a paper (2022) that images the gel formation in situ, and proposes simple models for the thickness of the gel layer. This is work by two postdocs, Ian Williams and Sara Naderizadeh.
Older work on paints
When an emulsion paint is applied to a wall, you are applying a thin film of liquid containing small (mostly less than a micrometre) particles. As the initially liquid film dries the water evaporates leaving these particles behind, and as the water leaves the particles are pushed together, and then these particles coalesce to form the final layer of paint.
A few years ago, a postdoc working with us, Andrea Fortini, studied the drying of a mixture of large (shown as red below) and small (yellow) particles. He found that if drying was fast enough, the large and small particles spontaneously segregated into a layer of the small particles on top of a layer of the larger particles (right-hand image). An experimental postdoc, Nacho Martin-Fabiani, then showed this also occurred in experiment, and we published both simulation and experiment in Phys Rev Lett paper (arXiv) in 2016.
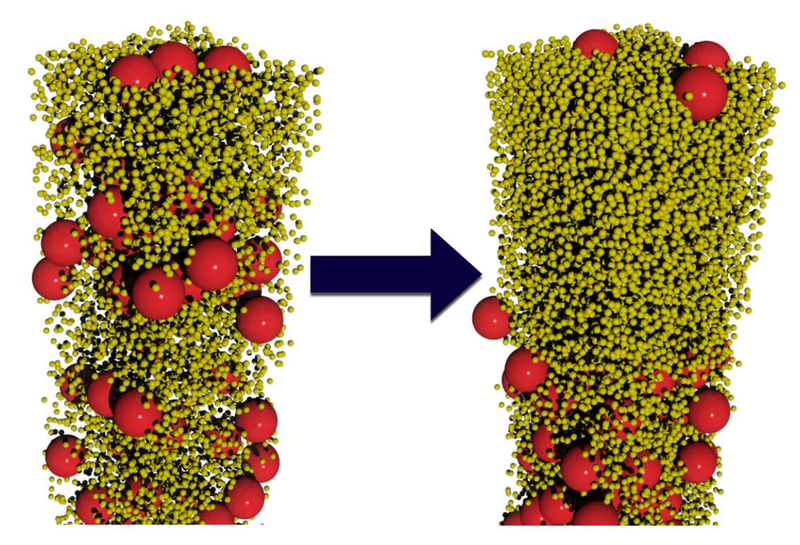
The work is described in a news item by the American Physical Society’s news service Physics. It has an embedded movie by Andrea if you want to see the effect happening. There is also a piece on it in the UK’s Institute of Physics’ Physics World.
In the Phys. Rev. Lett., Andrea and I also proposed a simple mechanism for the segregation of the small and large particles into layers. But this model neglected gradients in the pressure of the evaporating water. Patrick Warren and I have realised that this neglect is not justified; we have recently published a theory that does consider these gradients (arXiv). Our work also shows that the mechanism is an example of diffusiophoresis — the movement of one species (here the larger particles) in a concentration gradient of another species (the small particles.